Reputable suppliers can certify their raw materials through Informed Ingredient's raw material certification programme.
As part of the process, all ingredient batches are analysed in a specialised ingredient analysis laboratory to detect contamination at trace levels. The Informed Ingredient programme was created by LGC, an industry leader in quality assurance and testing of sports nutrition ingredients for World Anti-Doping Agency (WADA) banned substances. It enables ingredient suppliers to have confidence in both the ingredient and also in respect of the quality control procedures in place at the manufacturing facility to reduce the risk of contamination.
The Informed Ingredient raw material certification provides suppliers with the assurance that their ingredients carrying the Informed Ingredient mark have been regularly tested for substances considered prohibited in sport. In addition, Informed Ingredient also ensures that ingredients have been manufactured to high-quality standards.
Our raw material certification process ensures your ingredients meet global testing and quality benchmarks before reaching the marketplace. Click below to contact us and begin the path to Informed Ingredient certification.
How the Raw Material Certification Process Works
Certification with Informed Ingredient involves four stages:
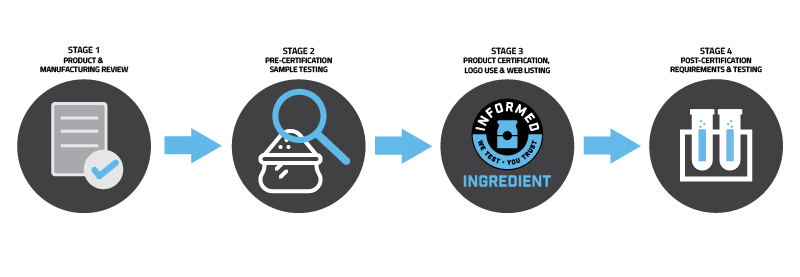
Stage 1: Ingredient & Manufacturing Review
A full review of the procedures in place at the manufacturing facility is undertaken via a paper-based assessment. This process is managed by experienced assessors, who provide guidance on how to meet the required standards of the Informed Ingredient programme and reduce risk within manufacturing processes.
These checks form the foundation of our raw material certification process:
- Quality Systems and Audits
- Staff Training in Relation to Cross-Contamination Prevention
- Raw Materials
- Raw Material Supplier Assessment Procedures
- Traceability & Recall Procedures
- Ingredient Contaminant Testing
Stage 2: Pre-Certification Sample Testing
Pre-certification involves testing a minimum of three pre-certification samples using LGC's ISO 17025 accredited prohibited substances supplement screen. All samples must consist of a minimum ingredient weight of 100g, either in final sealed packaging, or dispensed into a clean (unused) secure packaging. Analysis of these samples must not show evidence of contamination with prohibited substances.
This testing will be carried out in parallel with the Ingredient & Manufacturing Review (Stage 1) All analyses are conducted in an ISO 17025 accredited ingredient analysis laboratory, ensuring accurate and defensible results.
Download Our Certification Process Guide
Stage 3: Certification Completion, Logo Use and Web Listing
Once Stages 1 and 2 are complete and any resulting actions have been closed out, the ingredient will be accepted for certification with Informed Ingredient. An Informed Ingredient certificate will be issued, after which time the new Member company may use the Informed Ingredient logo in association with the certified ingredient. The Member company will be provided with the ‘Informed Ingredient Brand Standards’ which explains how the logo may be used and how best to market the new certification mark. The certified ingredient will be listed on the Informed Ingredient website along with all tested batches. A link to the Member company's website is also included.
Stage 4: Post-Certification Testing
Following certification, the Informed Ingredient is subjected to a regular testing process. As a standard process, each month the Member company will submit the latest production batch for testing using LGC's ISO 17025 accredited prohibited substances supplement screen. Companies may choose to test additional batches above and beyond the monthly testing regime or may test every batch if preferred. This allows Member companies to ensure the testing profile is fit for its purpose and meets the needs of their customers, whilst fulfilling the requirements of regular testing for banned substances.
Where a Member company can confirm a production frequency of fewer than 12 batches per year of the certified ingredient, the Member may elect to submit a sample of every production batch to LGC; samples being submitted immediately following production. Batch volumes and samples will be monitored to ensure the appropriate testing frequency is being adhered to. Our four-stage raw material certification framework is trusted by ingredient manufacturers worldwide.






